Computer Lab Semester - 5¶
Problem - 1¶
Solve the s-wave Schrödinger equation for the ground state and the first excited state of the hydrogen atom \begin{align} &\frac{d^2u}{dr^2}=A(r)u(r),\\ &A(r) = \frac{2m}{\hbar^2}[V(r)-E],\\ \mbox{where, } &V(r)=-\frac{e^2}{r} \end{align} where, m is the reduced mass of the electron. Obtain the energy eigenvalues and plot the corresponding wave functions. Remember that the ground state energy of the hydrogen atom is ≈-13.6 eV. Take e=3.795 (eVÅ), ħc= 1973(eVÅ) and m=0.511×10\(^6\) eV/c\(^2\).
Time independent Scrodinger equation¶
\begin{equation} \frac{d^2\psi(x)}{dx^2} + k^2(x)\psi(x) = 0 \end{equation} where, \begin{equation} k^2(x)=\frac{2m}{\hbar^2}(E-V(x)) \end{equation}
We split the second order differential equation into two first order equations as below - \begin{equation} \begin{aligned} &\frac{d\psi}{dx}=\phi(x)\\ &\frac{d\phi}{dx}=-k^2(x)\psi(x) \end{aligned} \end{equation}
[1]:
import numpy as np
import matplotlib.pyplot as plt
from scipy.integrate import odeint
from scipy.optimize import bisect
[2]:
# Basic parameters
E0 = -13.6 # approximate ground state energy
e = 3.795 # charge of electron in eVA
hbarc = 1973 # in eVA
m = 0.511e6 # in eV/c^2
# Based on the given parameters, calculate 2m/hbar^2c
C = 2*m/hbarc**2
[3]:
# Defining Helper Functions
def V(r):
return - e**2/r
def A(r):
return C*(V(r)-E)
def dzdr(z, r):
x, y = z
dxdr = y
dydr = A(r)*x
dzdr = np.array([dxdr, dydr])
return dzdr
def waveFunc(energy):
global sol
global E
E = energy
sol = odeint(dzdr, z[0], r)
return sol[-1, 0]
[13]:
# setting arrays for storing values
r = np.arange(1e-15, 8, 0.01)
z = np.zeros([len(r),2])
x0 = 0.001
y0 = 1
z[0] = [x0, y0]
[19]:
# Finding energy eigen values
Energy = np.linspace(-20, 0, 100)
waveFuncRight = np.array([waveFunc(Eval) for Eval in Energy])
sign = np.sign(waveFuncRight)
eigenValue = []
for i in range(len(sign)-1):
if sign[i] == - sign[i+1]:
en = bisect(waveFunc, Energy[i], Energy[i+1])
en = round(en,4)
eigenValue += [en]
print('Energy Eigen Values are \n'+str(eigenValue))
Energy Eigen Values are
[-13.7319, -3.4059, -0.7694]
[20]:
# Plotting wavefunctions
def plot():
fig, ax = plt.subplots(figsize=(8, 5))
# set the x-spine
ax.spines['left'].set_position('zero')
# turn off the right spine/ticks
ax.spines['right'].set_color('none')
ax.yaxis.tick_left()
# set the y-spine
ax.spines['bottom'].set_position('zero')
# turn off the top spine/ticks
ax.spines['top'].set_color('none')
ax.xaxis.tick_bottom()
plot()
color = ['red', 'blue', 'orange']
for i in range(len(eigenValue)):
waveFunc(eigenValue[i])
plt.plot(r, sol[:,0], color=color[i], label='n = '+str(i)+' E'+str(i)+' = '+str(round(eigenValue[i],1)))
plt.xlabel('r')
plt.ylabel('$\psi(r)$')
plt.legend()
#plt.grid()
plt.show()
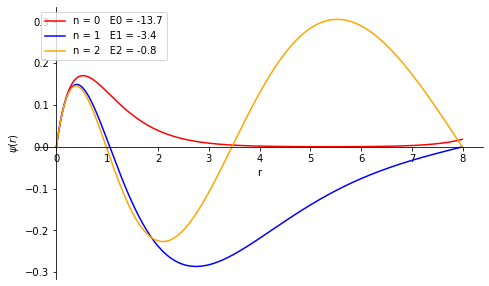
Problem - 2¶
Solve the s-wave radial Schrödinger equation for an atom \begin{align} &\frac{d^2y}{dr^2}=A(r)u(r),\\ &A(r) = \frac{2m}{\hbar^2}[V(r)-E],\\ \mbox{where, } &V(r)=-\frac{e^2}{r} \end{align} Where m is the reduced mass of the system (which can be chosen to be the mass of an electron), for the screened Coulomb potential \begin{align} V(r) = -\frac{e^2}{r}e^{-r/a} \end{align} Find the energy (in eV) of the ground state of the atom to an accuracy of three significant digits. Also, plot the corresponding wave function. Take e=3.795 (eVÅ), and a=3 Å, 5 Å, and 7 Å in the units of ħc = 1973(eVÅ) and m=0.511×10\(^6\) eV/c\(^2\). The ground state energy is expected to be above -12 eV in all three cases.
[21]:
import numpy as np
import matplotlib.pyplot as plt
from scipy.integrate import odeint
from scipy.optimize import bisect
[22]:
# Basic parameters
E0 = -12 # approximate ground state energy
e = 3.795 # charge of electron in eVA
hbarc = 1973 # in eVA
m = 0.511e6 # in eV/c^2
a = 5 # in units of angstrum
# Based on the given parameters, calculate 2m/hbar^2c
C = 2*m/hbarc**2
[23]:
# Defining Helper Functions
def V(r):
return - (e**2/r )*np.exp(-r/a)
def A(r):
return C*(V(r)-E)
def dzdr(z, r):
x, y = z
dxdr = y
dydr = A(r)*x
dzdr = np.array([dxdr, dydr])
return dzdr
def waveFunc(energy):
global sol
global E
E = energy
sol = odeint(dzdr, z[0], r)
return sol[-1, 0]
[24]:
# setting arrays for storing values
r = np.arange(1e-15, 10, 0.01)
z = np.zeros([len(r),2])
x0 = 0.001
y0 = 1
z[0] = [x0, y0]
[25]:
# Finding energy eigen values
Energy = np.linspace(-20, 0, 100)
waveFuncRight = np.array([waveFunc(Eval) for Eval in Energy])
sign = np.sign(waveFuncRight)
eigenValue = []
for i in range(len(sign)-1):
if sign[i] == - sign[i+1]:
en = bisect(waveFunc, Energy[i], Energy[i+1])
eigenValue += [en]
print('Energy Eigen Values are \n'+str(eigenValue))
Energy Eigen Values are
[-11.06410939502324, -1.284295766697573]
[26]:
# Plotting wavefunctions
def plot():
fig, ax = plt.subplots(figsize=(8, 5))
# set the x-spine
ax.spines['left'].set_position('zero')
# turn off the right spine/ticks
ax.spines['right'].set_color('none')
ax.yaxis.tick_left()
# set the y-spine
ax.spines['bottom'].set_position('zero')
# turn off the top spine/ticks
ax.spines['top'].set_color('none')
ax.xaxis.tick_bottom()
plot()
color = ['red', 'blue', 'orange']
for i in range(len(eigenValue)):
waveFunc(eigenValue[i])
plt.plot(r, sol[:,0], color=color[i], label='n = '+str(i)+' E'+str(i)+' = '+str(round(eigenValue[i],1)))
plt.xlabel('r')
plt.ylabel('$\psi(r)$')
plt.text(8,0.08, '$V(r)=\\frac{e^2}{r}e^{-r/a}; \\ a$ = '+str(a))
plt.legend()
#plt.grid()
plt.show()
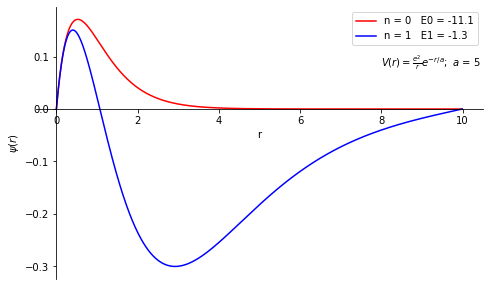
Problem 3¶
Solve the s-wave Schrodinger equation for the ground state and the first excited state of the hydrogen atom \begin{equation} \frac{d^2y}{dr^2}=A(r)u(r),\hspace{1cm}A(r)=\frac{2m}{\hbar^2}[V(r) - E]\mbox{~where~}V(r)=-\frac{e^2}{r} \end{equation} The anharmonic potential \begin{equation} V(r)=\frac{1}{2}kr^2 + \frac{1}{3}br^3 \end{equation} for the ground state energy (in MeV) of particle to an accuracy of three significant digits. Also, plot the corressponding wave function. Choose \(m=940 MeV/c^2\), \(k=100 MeVfm^{-2}\), \(b=0,10,30 MeVfm^{-3}\). In these units, \(c\hbar=940 MeV/c^2\), \(k=100 MeVfm^{-3}\) for all three cases. The ground state energy is expected to lie in between 90 and 110 MeV for all three cases.
[34]:
import numpy as np
import matplotlib.pyplot as plt
from scipy.integrate import odeint
from scipy.optimize import newton, bisect, brentq
from scipy.constants import hbar, m_e
[35]:
# Defining helper functions
# (1) Potential function
def V(r):
V = (1/2)*k*r**2 + (1/3)*b*r**3
return V
# (2) Defining A(r)
def A(r):
a = (2*m_e/ħc**2)*(V(r) - E)
return a
# (3) Defining Derivative functions of 'y'
def deriv(y, r):
return np.array([y[1], A(r)*y[0]])
# (4) Defining wave function with respect to energy
def waveFunc(energy):
global E
global psi
E = energy
psi = odeint(deriv, psi0, r)
return psi[-1, 0]
# (5) Finding zero of the wavefuncton by Shooting method
def zero(x, y):
energyEigenValue = []
s = np.sign(y)
#s = np.array(s, dtype=int)
for i in range(len(s)-1):
if s[i] == -s[i+1]:
zero = bisect(waveFunc, x[i], x[i+1])
energyEigenValue.append(zero)
return energyEigenValue
# Vectorizing the functions
V = np.vectorize(V)
waveFunc = np.vectorize(waveFunc)
zero = np.vectorize(zero)
[42]:
# Important parameters
m_e = 940
ħc = 197.3
k = 100
# Initialisation
N = 1000 # Number of points on x-axis
psi = np.zeros([N, 2]) # To store wave function values and its derivative
psi0 = np.array([0.001, 0]) # wavefunction of initial staes
b = 30
r = np.linspace(-3, 3, N) # x - axis
[43]:
En = np.arange(0, 150, 1)
psiRight = waveFunc(En)
#psiRight
[44]:
def zero(x, psi):
energyEigenValue = []
s = np.sign(psi)
#s = np.array(s, dtype=int)
for i in range(len(s)-1):
if s[i] == -s[i+1]:
zero = bisect(waveFunc, x[i], x[i+1])
energyEigenValue.append(zero)
return energyEigenValue
[45]:
EigenValue = np.array(zero(En, psiRight))
EigenValue
[45]:
array([ 31.55704024, 92.01776957, 144.81662013])
[46]:
fig = plt.figure(figsize=(5, 10))
fig.subplots_adjust(hspace=0.4, wspace=0.4)
j=1
for i in range(len(EigenValue)):
ax = fig.add_subplot(len(EigenValue), 1, j)
waveFunc(EigenValue[i])
plt.plot(r, psi.transpose()[0])
ax.spines['left'].set_position('zero')
ax.spines['right'].set_color('none')
ax.spines['top'].set_color('none')
ax.spines['bottom'].set_position('zero')
#plt.legend()
plt.xlabel('$r$', fontsize=14)
plt.ylabel('$\psi(r)$', fontsize=14)
j+=1
plt.show()
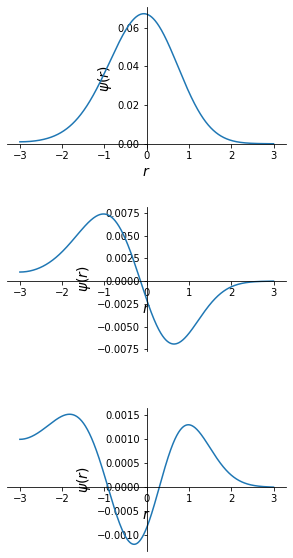
Problem 4¶
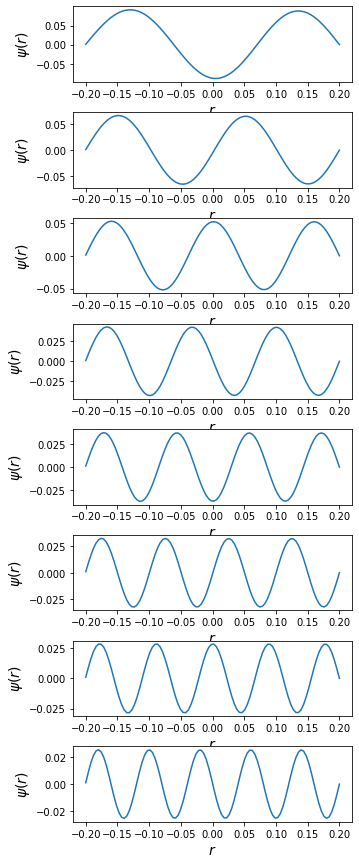
Solve the s-wave Schrodinger equation for the hydrogen molecule \begin{equation} \frac{d^2y}{dr^2}=A(r)u(r),\hspace{1cm}A(r)=\frac{2\mu}{\hbar^2}[V(r) - E] \end{equation} where, \(\mu\) is the reduced mass of the two-atom system for the Morse potential. \begin{equation} V(r)=D\left(e^{-2\alpha r^{\prime}} - e^{-\alpha r^{\prime}}\right),\hspace{0.5cm}r^{\prime}=\frac{r - r_0}{r} \end{equation} Find the lowest vibrational energy (in MeV) of the molecule to an accuracy of three significant digits. Also plot the corressponding wave function. Take \(m=940\times 10^6 eV/c^2\), \(D=0.755501\), \(\alpha=1.44\) and \(r_0=0.131349 \overset{\circ}{A}\)
Morse potential expression given is wrong. The correct form is \begin{align} V(r) &= D \left[1 - e^{-\alpha(r-r_0)}\right]^2\\ V(r) &=D\left(1 - 2e^{-\alpha r^{\prime}} + e^{-2\alpha r^{\prime}}\right), \hspace{0.5cm} r^{\prime}=\frac{r - r_0}{r} \end{align}
[27]:
import numpy as np
import matplotlib.pyplot as plt
from scipy.integrate import odeint
from scipy.optimize import newton, bisect, brentq
from scipy.constants import hbar, m_e
[28]:
# Defining helper functions
# (1) Potential function
def V(r, r0, α, D):
V = D*(1 - np.exp(-α*(r - r0)))**2
return V
# (2) Defining A(r)
def A(r):
a = (2*μ/ħc**2)*(V(r, r0, α, D) - E)
return a
# (3) Defining Derivative functions of 'y'
def deriv(y, r):
return np.array([y[1], A(r)*y[0]])
# (4) Defining wave function with respect to energy
def waveFunc(energy):
global E
global psi
E = energy
psi = odeint(deriv, psi0, r)
return psi[-1, 0]
# (5) Finding zero of the wavefuncton by Shooting method
def zero(x, y):
energyEigenValue = []
s = np.sign(y)
#s = np.array(s, dtype=int)
for i in range(len(s)-1):
if s[i] == -s[i+1]:
zero = bisect(waveFunc, x[i], x[i+1])
energyEigenValue.append(zero)
return energyEigenValue
# Vectorizing the functions
V = np.vectorize(V)
waveFunc = np.vectorize(waveFunc)
zero = np.vectorize(zero)
[29]:
# Important parameters
μ = 940e6
D = 0.755501
α = 1.44
r0 = 0.131349
ħc = 1973
# Initialisation
N = 100 # Number of points on x-axis
psi = np.zeros([N, 2]) # To store wave function values and its derivative
psi0 = np.array([0.001, 2]) # wavefunction of initial staes
r = np.linspace(-0.2, 0.2, 100) # x - axis
[30]:
En = np.arange(0, 15, 1)
psiRight = waveFunc(En)
#psiRight
[31]:
def zero(x, psi):
energyEigenValue = []
s = np.sign(psi)
#s = np.array(s, dtype=int)
for i in range(len(s)-1):
if s[i] == -s[i+1]:
zero = bisect(waveFunc, x[i], x[i+1])
energyEigenValue.append(zero)
return energyEigenValue
[32]:
EigenValue = np.array(zero(En, psiRight))
EigenValue
[32]:
array([ 1.21457505, 2.10732938, 3.2543345 , 4.65597459, 6.31235437,
8.22351125, 10.389463 , 12.81021612])
[33]:
fig = plt.figure(figsize=(5, 15))
fig.subplots_adjust(hspace=0.4, wspace=0.4)
j=1
for i in range(len(EigenValue)):
ax = fig.add_subplot(len(EigenValue), 1, j)
waveFunc(EigenValue[i])
plt.plot(r, psi.transpose()[0])
#plt.legend()
plt.xlabel('$r$', fontsize=14)
plt.ylabel('$\psi(r)$', fontsize=14)
j+=1
plt.show()
[ ]: